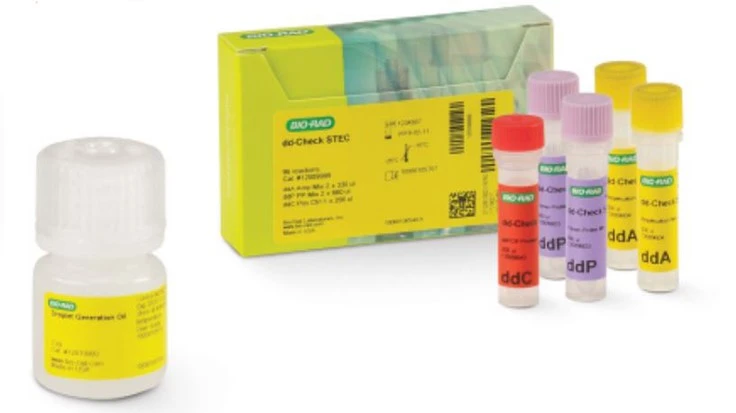
Hercules, Calif,-based Bio-Rad Laboratories, Inc. has launched dd-Check STEC Solution for detecting virulence genes from Shiga toxin-producing Escherichia coli (STEC). Pathogenic STEC is defined as the presence of both stx1 and stx2, as well as eae, in one E. coli bacterium. The kit, which harnesses Bio-Rad’s Droplet Digital PCR (ddPCR) technology, determines the coexistence of the STEC virulence genes in a single bacterium without additional confirmatory steps, providing rapid and reliable confirmation of presence of pathogenic STEC, and returning results in a few hours, the company said. Decreasing screening and confirmation turnaround time means products can be released to market faster, which is especially important for food matrices with limited shelf life, it added.
The dd-Check STEC Solution uses ddPCR technology to generate thousands of uniform nanoliter-sized droplets. Once generated, the droplets can be amplified, read, and analyzed for the colocalization of virulence genes by Bio-Rad Qx IDE Manager Software. By encapsulating a single bacterium in each droplet, the dd-Check STEC Kit can distinguish between a true positive and a negative result. As regulations classifying STEC as an adulterant continue to change, the kit provides the flexibility to adapt to new requirements. If other virulence factors are required for testing, the workflow can be updated to include additional testing.
To learn more about the dd-Check STEC Solution, visit bio-rad.com/ddcheck.
Latest from Quality Assurance & Food Safety
- FDA Releases Produce Regulatory Program Standards
- Invest in People or Risk the System: Darin Detwiler and Catalyst Food Leaders on Building Real Food Safety Culture
- USDA Proposes Increasing Poultry, Pork Line Speeds
- FDA Releases New Traceability Rule Guidance
- TraceGains and iFoodDS Extend Strategic Alliance
- bioMérieux Launches New Platform for Spoiler Risk Management
- SafetyChain Receives SOC 2 Type 2 Certification
- Puratos Acquires Pennsylvania-Based Vör Foods





